
Introduction
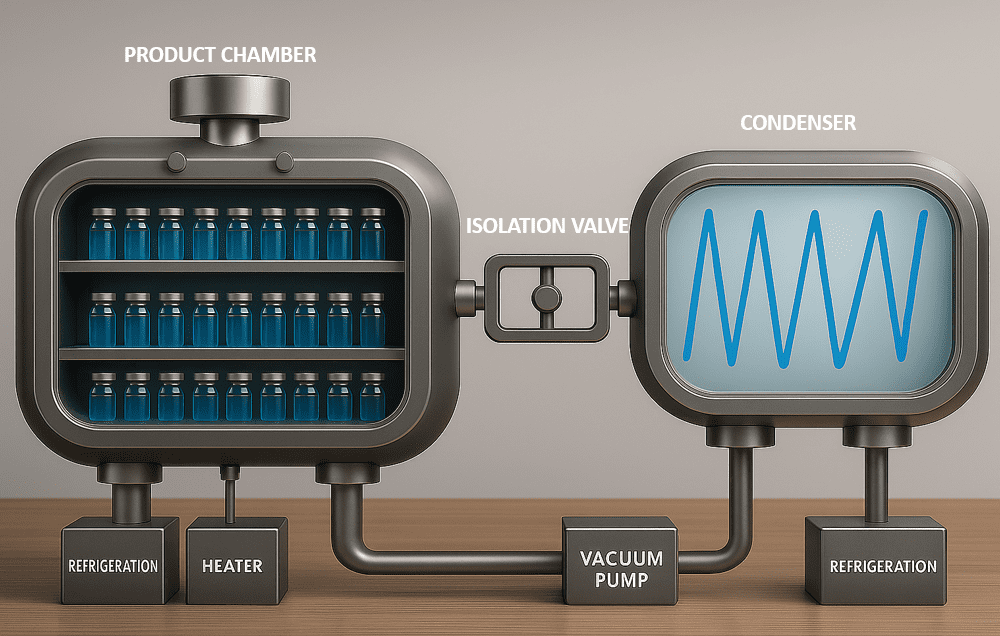
A modern lyophilizer comprises a vacuum‑sealed chamber with multiple heated shelves, a condenser unit, vacuum pump, and a PID-based controller. Products are loaded onto shelves, frozen to well below their collapse temperature (typically –40 °C to –80 °C), then subjected to sublimation under controlled heat and pressure ramps. Final secondary drying raises shelf temperature (e.g. 20–30 °C) to remove bound moisture—all under tightly regulated conditions.
Challenges
Thermally sensitive biopharmaceuticals risk structure collapse if product temperature exceeds its critical threshold (e.g. glass-transition temperature ~ –14 °C).
- Inconsistent moisture content across vials due to uncontrolled freezing and drying, leading to batch rejects.
Scale-up discrepancies—lab-scale cycles failing to translate reliably to pilot or production units, due to variations in Kv (heat transfer coefficient), ice nucleation, and heat flux.
Pressure control issues during mid-primary drying leading to uneven sublimation rates requiring valve tuning and PID adjustments.
Solution
GIC Digital PID Temperature Controller
- Sensor Type: RTD/PT100 or thermocouple for shelf and product temperature
- Control Mode: Closed‑loop PID via SSR or valve output
- Set Points (SV):
- Freezing: –40 °C to –80 °C
- Primary drying: ~4 °C to just below Tc (e.g. –15 °C)
- Secondary drying: ~20–30 °C
The PID controller adjusts heating (via shelf thermal fluid or heating elements) and vacuum/regulator valves in real time, based on sensor feedback. Ramp‑soak profiling ensures temperatures remain safely below collapse thresholds while maximizing drying rate.
Process Flow
1) Freezing:
Inside the product chamber, the shelf temperature is lowered until the solvent freezes, in this case the water is the solvent. The product has lower freezing point hence the temperature is further lowered to completely freeze the solution.
While the product was freezing the condenser was being cooled to well below the shelf temperature.

2) Primary drying phase.
The isolation valve is opened and a vacuum pump pulls out air of both the chambers. Once the pressure has decreased the temperature of the shelves is slowly increased.
The warmer temperature and lower pressure causes the ice crystal to turn directly to vapor a process called as sublimation. The water vapor rises up and out of the vial, leaving behind the partially dried product matrix.
The water vapor travels through the isolation valve and gets trapped on cold condenser coil. The vapor naturally moves from area of higher pressure where the sublimation occurs to the area of lower pressure near the frozen condenser coil.
By end of primary drying 90 % of water has been removed but it’s not completely dry yet because there’s still some water bound to the product
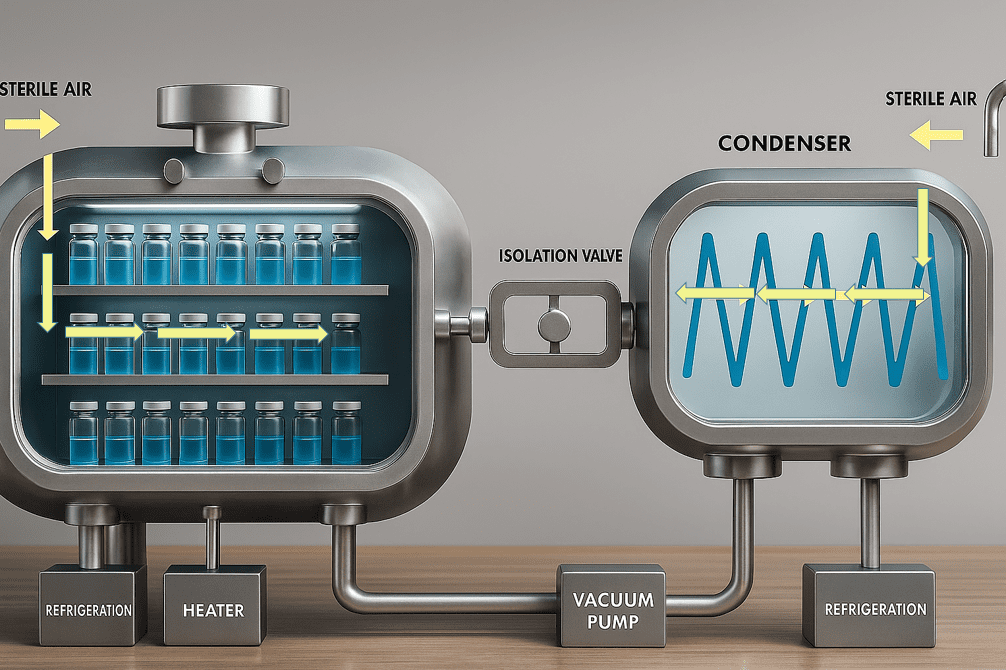
3. Secondary Drying:
Removing the bound water needs more heat.
So in secondary drying the temperature is slowly increased. This removes any remaining water that’s bound to the product matrix.
Leaving only the completely dried product matrix

4. Back filling:
After the product has dried sterile air is back filled into both the chambers to remove vacuum and bring it back to normal pressure.

Conclusion
A digital PID temperature controller integrated into a lyophilizer enables high-precision thermal regulation, uniform drying, and scalable freeze‑drying processes for sensitive formulations. The outcome is improved product quality, process reproducibility, and operational reliability—setting a benchmark for lyophilization performance.




